I’ve wondendered before, why is methamphetamine an illegal drug in most countries while dextroamphetamine is legal (although with strict controls) for treating conditions like ADHD?
If I look at a picture of these molecules, they are basically identical except for the extra n-methyl (NH₃) group found in methamphetamine:
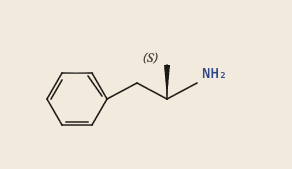
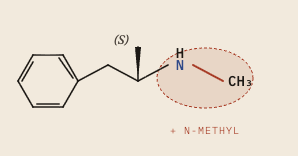
Their molecular formula is almost the same:
- C₉H₁₃N - Dextroamphetamine
- C₁₀H₁₅N - Methamphetamine
That extra n-methyl group is what causes methamphetamine to be so potent: it raises lipophilicity which means it methamphetamine corosses the blood-brain barrier faster, producing a shaper CNS (Central Nervous System) rise per dose. Methamphetamine is more strongly dopaminergic in the CNS, therefore it carriers higher abuse liability.
Note on strength comparison: in terms of mg per mg strength, I haven’t found concrete answers and it seems to depend on the the effect measured:
- Therapeutic ADHD
- Subjective CNS effects (euphoria, focus intensity)
- Pheripheral cardiovascular efffects
Most of the literature says 1:1 to 1:2 depending on what’s being measured.
AI generated summaries
The paragraphs below were written by Opus 4.7 and further validated by:
- Opus 4.7 (new session, no previous context)
- Opus 4.6 (new session, no previous context)
- ChatGPT 5.4
Shared mechanism
Both molecules act as substrate-type releasers at the plasma-membrane monoamine transporters (DAT and NET), inducing reverse transport of cytosolic dopamine and noradrenaline rather than blocking reuptake in the manner of cocaine or methylphenidate (Sulzer et al. 2005). The cytosolic monoamine pool that supplies this efflux is generated upstream by disruption of the proton gradient across synaptic vesicles, dissipating VMAT2-mediated sequestration (Sulzer et al. 2005). Both compounds also act as agonists at trace amine-associated receptor 1 (TAAR1) (Bunzow et al. 2001), a pathway that modulates DAT phosphorylation and dopaminergic firing and is now considered integral to amphetamine-class pharmacology (Miller 2011).
Where molecules diverge
The N-methyl substitution that distinguishes methamphetamine from dexamphetamine increases lipophilicity and accelerates blood–brain barrier penetration, producing a faster CNS onset at equivalent oral doses (Kirkpatrick et al. 2012). Methamphetamine is partly N-demethylated to amphetamine in vivo via CYP2D6, so a fraction of its later effect is amphetamine-mediated (Lin et al. 1997; Cruickshank and Dyer 2009). The reported elimination half-life of methamphetamine is approximately 9–12 hours with substantial inter-individual variability (Cruickshank and Dyer 2009), while dexamphetamine half-life is reported in the range of 10–14 hours and is sensitive to urinary pH due to substantial renal excretion of unchanged drug.
On the comparative human evidence
In a within-subject human study comparing intranasal d-methamphetamine and d-amphetamine, Kirkpatrick et al. (2012) found the two drugs produced similar dose-related profiles of subjective and physiological effects, with methamphetamine generating modestly greater effects on selected mood and cardiovascular measures and equivalent self-administration — consistent with comparable abuse potential when route is held constant. Clinical practice has long observed a roughly 1:1.5 to 1:2 oral-dose ratio between methamphetamine and dexamphetamine for ADHD treatment, reflected in the maximum approved daily doses of Desoxyn and Dexedrine in their respective FDA prescribing information.
References
- Bunzow JR, Sonders MS, Arttamangkul S, Harrison LM, Zhang G, Quigley DI, Darland T, Suchland KL, Pasumamula S, Kennedy JL, Olson SB, Magenis RE, Amara SG and Grandy DK (2001) ‘Amphetamine, 3,4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor’, Molecular Pharmacology, 60(6):1181–1188, doi:10.1124/mol.60.6.1181 Cruickshank CC and Dyer KR (2009) ‘A review of the clinical pharmacology of methamphetamine’, Addiction, 104(7):1085–1099, doi:10.1111/j.1360-0443.2009.02564.x
- Kirkpatrick MG, Gunderson EW, Johanson CE, Levin FR, Foltin RW and Hart CL (2012) ‘Comparison of intranasal methamphetamine and d-amphetamine self-administration by humans’, Addiction, 107(4):783–791, doi:10.1111/j.1360-0443.2011.03706.x
- Lin LY, Di Stefano EW, Schmitz DA, Hsu L, Ellis SW, Lennard MS, Tucker GT and Cho AK (1997) ‘Oxidation of methamphetamine and methylenedioxymethamphetamine by CYP2D6’, Drug Metabolism and Disposition, 25(9):1059–1064
- Miller GM (2011) ‘The emerging role of trace amine-associated receptor 1 in the functional regulation of monoamine transporters and dopaminergic activity’, Journal of Neurochemistry, 116(2):164–176, doi:10.1111/j.1471-4159.2010.07109.x
- Sulzer D, Sonders MS, Poulsen NW and Galli A (2005) ‘Mechanisms of neurotransmitter release by amphetamines: a review’, Progress in Neurobiology, 75(6):406–433, doi:10.1016/j.pneurobio.2005.04.003